Where Innovation Meets Regulatory Confidence
Where innovation moves fast but compliance never falls behind — empowering life sciences and healthcare organizations to advance research, protect patients, and operate with complete regulatory confidence.
The Operational Reality
Life Sciences organizations operate in highly regulated environments. But compliance, clinical operations, and quality workflows often run in disconnected systems. Without unified oversight, traceability becomes manual — and operational visibility stays limited.
SOP & Regulatory Governance
Policies and SOPs live in separate systems. Traceability depends on manual coordination.
Clinical & Research Coordination
Clinical activities generate critical data. Visibility across teams is limited.
Vendor & CRO Oversight
External partners operate in silos. Oversight is fragmented and reactive.
Quality Events & CAPA Management
Incidents and deviations are logged. But root causes and corrective actions lack full lifecycle context.
Regulated Workforce & Training
Training and compliance tracking run separately from execution. Audit readiness becomes a project — not a built-in capability.
The Missing Architecture
Life Sciences operates under strict regulation. But systems rarely provide a unified operational structure. What’s missing isn’t another tool — it’s a connected governance architecture.
What It Takes to Operate Under Regulation — Intelligently
Regulated operations don’t fail because of effort. They fail when compliance, clinical, quality, and operational data operate in silos.
Operating under regulation requires a governed, unified foundation — where traceability, validation, and intelligence coexist.
Why CloudOffix?
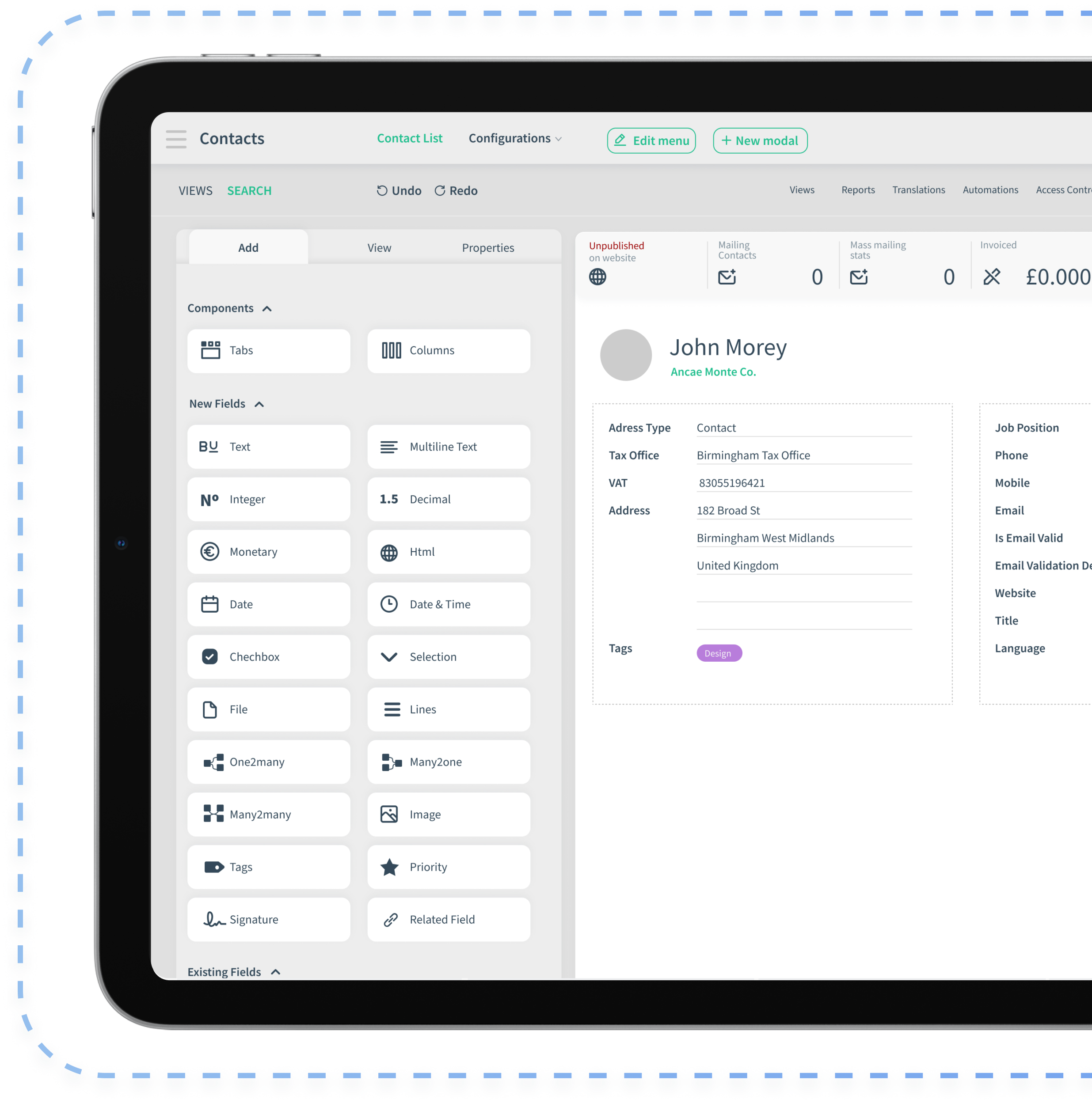
CloudOffix doesn’t replace EHR, EMR, or clinical systems.It unifies regulated execution — from SOP governance to quality oversight.
SOP Governance
Policies and changes flow through structured, traceable workflows.
Clinical & Research Coordination
Activities, documents, and milestones connect in one system.
Vendor & CRO Oversight
Contracts, risks, and performance stay visible across partners.
Quality Events & CAPA
Deviations, audits, and CAPAs run in one traceable flow. Issues are detected early and resolved systematically.
Training & Certification Management
Training status, certifications, and renewals stay aligned with roles. No gaps. No last-minute compliance risks.
Audit & Inspection Readiness
Every action is logged and audit-ready. Inspection data is structured — not assembled manually.
Business Impact
Accelerate Audit Readiness
Audit prep becomes continuous — not reactive. Inspection confidence increases.
Reduce Compliance & Operational Risk
Risks surface early across vendors, processes, and certifications. Control replaces surprises.
Improve Clinical & Project Execution Speed
Milestones and escalations stay structured and visible. Execution becomes measurable.
Strengthen Vendor & CRO Oversight
Contracts, SLAs, and performance tracked in one system. Oversight becomes proactive.
Increase Organizational Transparency
Leadership sees live compliance, training, and quality signals. Decisions move from static reports to real-time insight.
Lower Total Operational Cost of Compliance
No manual prep. No duplicated data.Less friction. Lower long-term compliance cost.
AI That Understands Your Entire Agency
Most AI tools operate on fragments. CloudOffix AI operates on the full engagement-to-revenue lifecycle.
AI That Understands Regulation
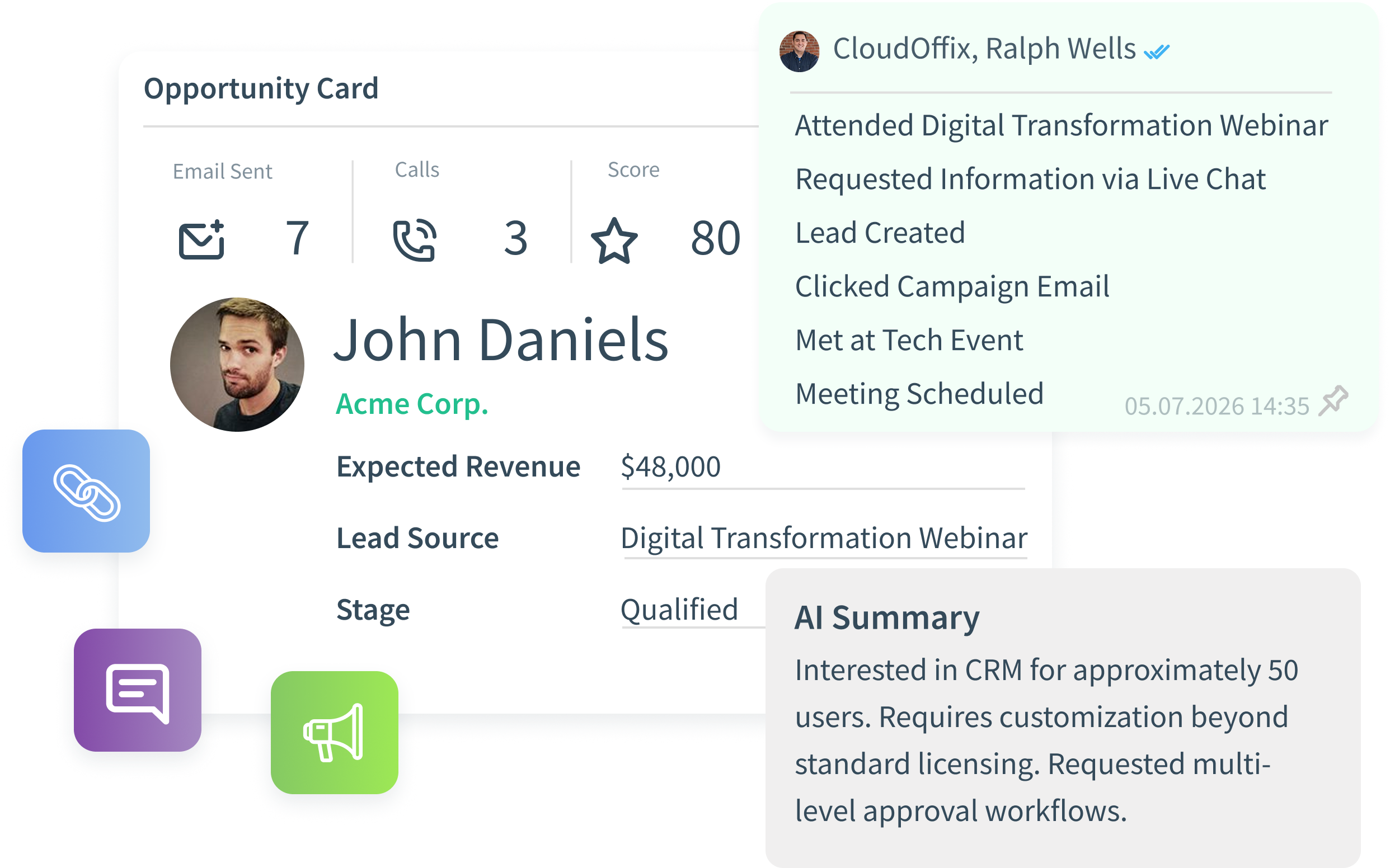
Most AI in Life Sciences sits outside regulated workflows. CloudOffix AI operates inside execution — across SOPs, clinical coordination, vendor oversight, quality events, and training. It runs on structured, governed data. It analyzes traceable operational reality. This is controlled operational intelligence — not experimental AI.






Ready to Operate With Confidence Under Regulation?
Regulated performance requires visibility and control. With governed operational data, organizations gain:
• Faster audits
• Earlier risk detection
• Stronger compliance
• More predictable execution
Compliance becomes continuous. Intelligence becomes embedded.